ClO2 is known for its antimicrobial, antiviral, and antifungal activity when applied in controlled range of concentrations between 5 and 8000 ppm1,23,24,28. Numerous researchers explored its potential in in vitro, in vivo studies and clinical trials17,24,25,26,27,28,29,30,31,32,33,34,35. Also advocates of MMS assert its capability to treat a range of illnesses. However, such claims lack empirical support regarding both its safety and therapeutic effectiveness.
Herein, we aimed to investigate antimicrobial/antibiofilm activity and cytotoxicity of ASC, a source of ClO2 (Tables 1, 2, 3 and 4, Figs. 1, 2, 3 and 4). In our study, ClO2 was obtained by acidifying SC using either an inorganic or an organic acid (ASC1, ASC2, respectively).
The lowest MIC of 0.002992% (29.92 ppm) was observed against MSSA and E. coli when ASC2 was applied. For ASC1, the same MIC value of 0.002992% (29.92 ppm) was observed only against MSSA (Table 1). Both formulations at this concentration showed acceptable levels of cytotoxicity but did not exhibit stronger antimicrobial activity than PHMB (the clinically used antiseptic agent) or the 5 and 20 ppm ASC solutions used by Ma et al.28 (Table 4). The discrepancies observed may be related to either the different strains tested in this work or the ClO2 production method, as the electrolytic method reduces impurities in the solution, potentially affecting its antimicrobial efficacy28,34.
Conversely, MIC of PHMB was higher than ASC2 towards P. aeruginosa and L. casei and of ASC1 towards P. aeruginosa. Our results are consistent with results of Morino et. al., which have shown the effectiveness of extremely low concentration (0.01 ppm, 0.028 mg/m3) ClO2 against P. aeruginosa and E. coli36. In the case of E. coli and L. casei, ASC2 displayed a higher antimicrobial effect than ASC1. This may indicate that gluconic acid may have an additional role in the antibacterial effect.
Björnsdóttir et al. have used gluconic acid as a non-inhibitory low-pH buffer while investigating the acid-resistance of E. coli, however Eisenberg et al. revealed that gluconate induces the Entner-Doudoroff pathway activity and catabolism of glucose in E. coli which could additionally disturb the functioning of bacterial cell and reduce its viability37,38. Gluconate-induced pathway of glucose catabolism in L. casei is unconfirmed as far. The observed reduction in L. casei number may indicate that when drinking, ASC solution may disturb the gut microbiome and speculatively vaginal microbiota as well. All biofilms were more tolerant to applied compounds than their planktonic counterparts (Tables 1 and 2)39,40. S. mutans and E. coli biofilms were the most susceptible to ClO2. Accordingly, Herczegh et al. showed that 0.0015% and 0.003% solutions of hyper-pure ClO2, obtained with dedicated methods, are promising preventive and therapeutic adjuvants in dental practice as they reduced the number of S. mutans in saliva samples collected from patients41. The susceptibility of E. coli biofilm to ASC2 may be related to the activation of the Entner-Doudoroff pathway as mentioned above38.
SC was more effective against the biofilm of S. mutans than ASC1 and ASC2 (Table 2, Fig. 2), which stays in line with reports showing that biofilm formation and matrix gene transcription can be stimulated by sublethal (4 μg/ml; 150 ppm) doses of ClO242,43.
It was shown that L. rhamnosus biofilm displayed high susceptibility to sub-MBEC (0.09575%, 957.5 ppm) which may suggest that consuming even low amount of ClO2 may interfere negatively with microbiota (Fig. 2). However, our results encroach on Ahmed et al. observations indicating that ClO2 in concentrations 0.05%; 500 ppm and 0.1%; 1000 ppm did not affect the Lactobacillus spp.44.
The different species within Lactobacillus genera display various susceptibility to 75 ppm and 125 ppm ASC which have been explained by various adaptations to fermentative process conditions and so, different resistance to antimicrobial agents45.
The certain discrepancy observed at MBEC (Fig. 2), where green-dyed cells remain in the fluorescence microscopy data despite complete eradication of biofilms observed in the 96-well microplate model (Table 3), may be partially attributed to the differences in the experimental setup. The microplate model utilizes a 96-well plate format, while the fluorescence microscopy was conducted in a 24-well plate. Although the same concentrations of ASC1 and ASC2 were applied in both setups, scaling these concentrations 1:1 between the two models may only serve as an estimation rather than a precise match. Differences in the surface area-to-volume ratios, diffusion dynamics, and overall biofilm density between the 96-well and 24-well plates could influence how the antiseptics interact with the biofilms46. In particular, the larger well size in the 24-well plate may result in more uneven distribution of the antiseptic or altered biofilm growth patterns, allowing some cells to survive. Nonetheless, despite these differences, the results from both models were largely consistent, with the fluorescence microscopy data supporting the findings from the microplate model to a significant extent. This consistency reinforces the validity of the MBEC values derived from the microplate model, though it also highlights the need to account for such scaling factors when comparing data across different experimental platforms.
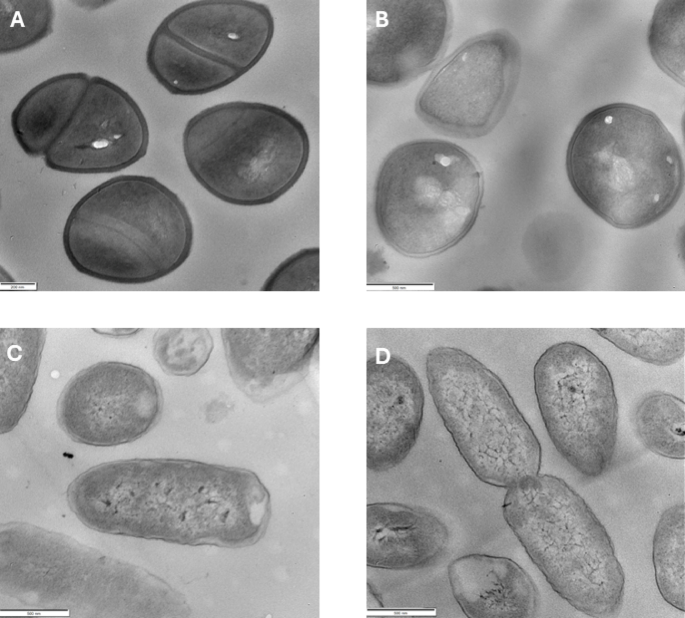
S. mutans biofilm was also the most resistant to ASC1 in the investigation of biofilm eradication from hydroxyapatite surface investigation (Fig. 3A–C). It is known that S. mutans can grow in continuous cultures in pH values of 4.5–5.0 however, this microorganism can survive brief periods of extreme acidification (pH 2.5). Gram-positive microorganisms demonstrated higher resilience to ClO2 than Gram-negative ones (Fig. 3) which may be explained by the enhanced stability of the membrane structure and the mechanical stability of their cell walls47,48. A high resistance to ClO2 (0,03%) was also described in the case of other Gram-positive Bacillus subtilis vegetative forms isolated from a washer‐disinfector49.
The biofilm formed by E. coli and L. casei was the most susceptible to ASC1 (Fig. 3B, C). Although ASC1 is effective against biofilm formed by Gram-negative species, a higher eradication effect was noted against E. coli than P. aeruginosa biofilm, confirming the results obtained in previous methods and proving that bacterial species differ in susceptibility to antimicrobials even if they share similar cell structures that form cell wall (Table 2, Figs. 2 and 3B). However, a relatively high level of biofilm eradication was observed also in the case of P. aeruginosa biofilm which is opposite to MBEC results. This may be related to the structure of the biofilm formed by this species. P. aeruginosa biofilm consists of densely packed populations of cells embedded in tick, mucosal extracellular polymeric substances (EPS)50. This can affect the detachment of large cell aggregates from the structure of the biofilm during the procedure performance. Therefore, the results obtained in this step of investigation may be burdened with a bias arising from the methodology applied51. The fact that L. casei was eradicated more effectively from HAP surface than L. rhamnosus biofilm, confirms that the intrinsic susceptibilities of microorganisms to disinfectants vary widely [B]. Nevertheless, all tested microorganisms formed robust biofilm structures on HAP surfaces (Fig. 4) what may satisfactorily explain the high differences between recorded MIC and MBEC values.
It is believed that the duration of acidification of SC has an impact on its biological activity3. In this study, we have shown that after 180 s of acidification, effective concentrations of ASC were 2—8 times lower against MSSA and P. aeruginosa biofilms, respectively, compared to the situation when 60 s of acidification was applied. This result may influence the production of ASC solution for medical purposes20,21,22,23,24. To make ClO2, an acid is mixed with chlorite, which slowly releases the gas. The reaction normally requires high acidity (low pH) which is irritating for human tissues. Also in this study, we have observed a concentration-related increase of cytotoxic effects in both in vitro and in vivo analysis (Table 4, Fig. 5) after the use of antimicrobially-active ASC concentrations. While the G. mellonella model is a widely recognized model for toxicity evaluation, methodological parameters implemented into this research provide data on subchronic toxicity the limitation of this study is the absence of long-term toxicity assessments on mammalian cells52,53,54. Therefore, the use of obtained cytotoxicity data should be approached with caution due to physiological differences between insect and mammalian systems. A robust summary concerning the overall perspective on the toxicology of chlorine dioxide and chlorite is available on the Agency for Toxic Substances and Disease Registry website (Atlanta, USA)55. Nevertheless, based on the data obtained it may be hypothesized that choosing mild acid with a pH closer to neutral and prolonged acidification time may result in a product optimized for use on the body. Interestingly, the rapid death (within 2 h) of larvae after application of ASC (Fig. 5) was effect even harsher than level of cytotoxicity observed after exposure of fibroblast cell line to ASC.
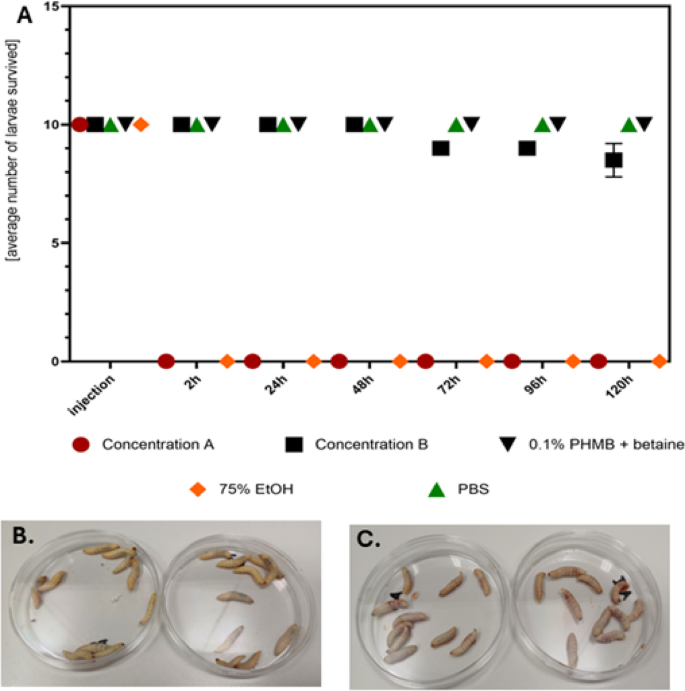
(A) Average survival of larvae treated with concentration A (0.383% ClO2) or B (0.002992% ClO2) of ASC1, PHMB, PBS, and 70% ethanol (B) larvae treated with concentration B of ASC1 display full turgor, mobility and viability. (C) Larvae treated with concentration A of ASC1 after two hours stopped moving and started to vomit red-colored liquid. Within the next hours these larvae became melanized and did not react to physical stimuli. Average [%] survival of larvae treated using concentration A or ethanol already after 1 h post-exposure was 0%; [%] survival of larvae treated using concentration B after 120 h was 75%]; [%] survival of larvae treated using 0.1% PHMB + betaine or PBS was 100%.
Our results indicate that it is difficult to establish a precise antimicrobial dose of ASC while being effective against pathogens, safe for tissues and sparing probiotic species. We have shown that the dosage recommended by alternative medicine proponents cannot lead to the achievement of safe and therapeutic concentrations. At the same time, taking an ASC solution with a pH of 2.5 several times a day can led to damage to the mucous membrane of the gastrointestinal tract. Taking into consideration existing research results concerning ClO2 it can be noted that it has the potential to be used for medicinal purposes, but more investigation is needed to develop safe methods of its application and to evaluate a factual benefit/risk ratio.
link