An article published in the Journal of Controlled Release details the results of a cross-border scientific collaboration that has developed an alternative for the treatment of solid tumors based on inhibition of the inflammatory tumor microenvironment (TME).
Solid tumors are difficult to treat, partly owing to cells and substances in the TME that prevent drugs from penetrating and stop the patient’s own immune system from combating the tumor. “These cells and molecules often fuel the growth of the tumor, which escapes immune surveillance,” said Lúcia Helena Faccioli, a co-author of the article. She is a professor at the University of São Paulo’s Ribeirão Preto School of Pharmaceutical Sciences (FCFRP-USP) in Brazil and heads the Center of Excellence for Quantification and Identification of Lipids (CEQIL), established with FAPESP’s support via its Multiuser Equipment Program.
“Immune cells that promote and inhibit tumors tend to be always in a tug-of-war in the TME, where metabolites, lipid mediators, cytokines and chemokines play an important role in the dominance of immunosuppressive nature,” the article states.
The other co-authors include Viviani Nardini, a researcher in FCFRP-USP’s Department of Clinical, Toxicological and Bromatological Analysis, and scientists affiliated with Indian institutions, led by Avinash Bajaj, head of the Nanotechnology and Biological Chemistry Laboratory at the Regional Center for Biotechnology in Faridabad, Haryana state.
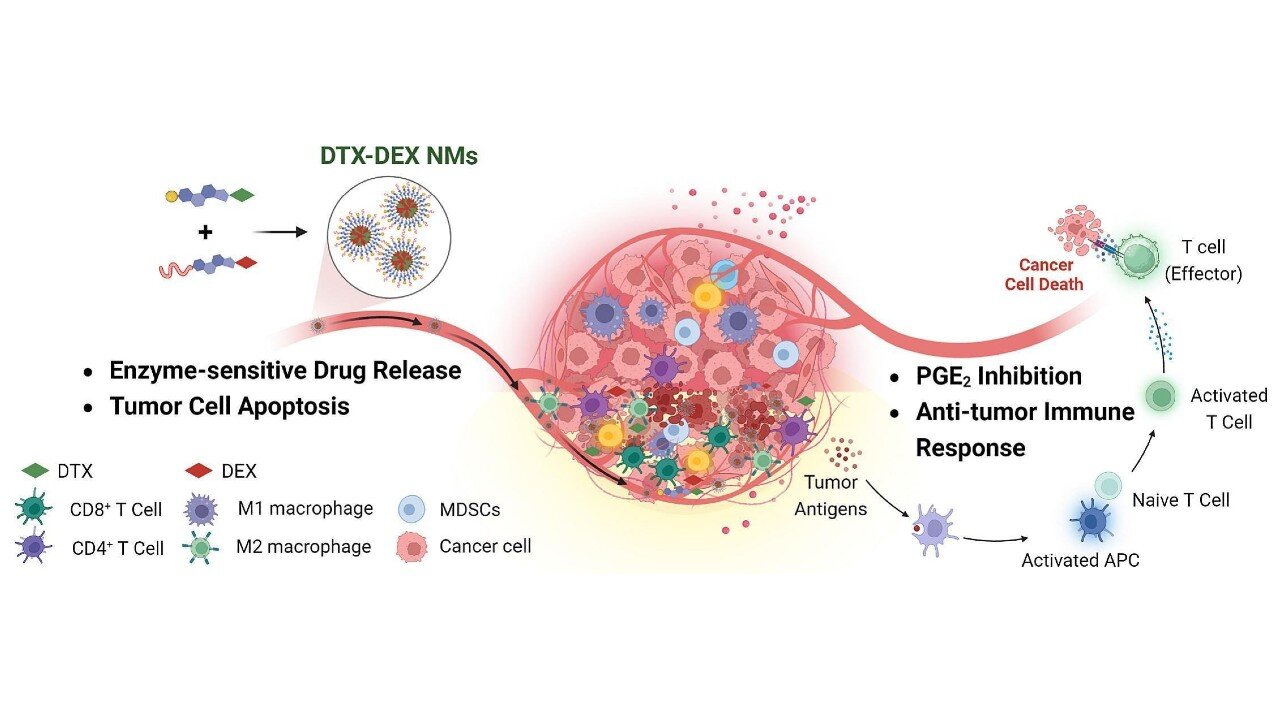
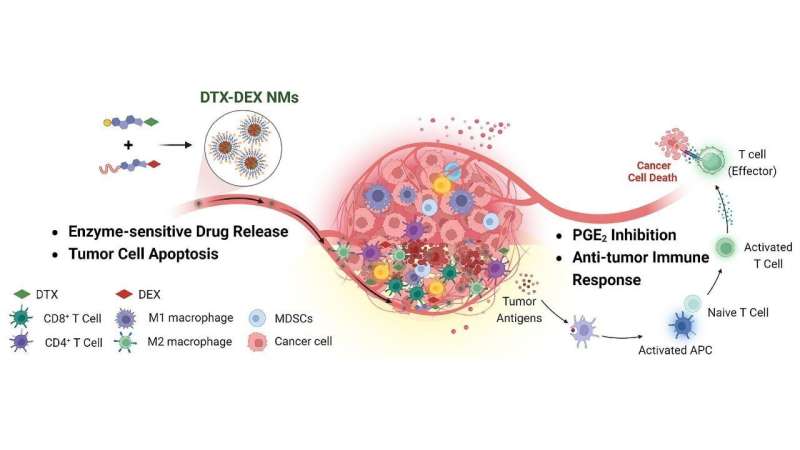
The team developed nanomicelles (very small particles, measuring between 1 and 100 nanometers) comprising different substances and for this reason termed chimeric. The chimeric nanomicelles were composed of phospholipids, docetaxel (DTX), a chemotherapy medication that destroys tumor cells, and dexamethasone (DEX), an anti-inflammatory drug widely used to reduce production of various inflammatory substances, such as prostaglandin E2 (PGE2).
Tests in laboratory animals showed that these DTX-DEX NMs were highly effective when administered intravenously, reducing tumor size and increasing survival. Untreated animals always died aged 28–30 days, while treated animals survived to age 44–50 days, according to Faccioli.
“The treatment induced a reduction of more than five times in tumor volume compared with untreated tumors in the colon cancer model,” Bajaj said. The nanomicelles reduced and altered the cells around the tumor that prevent action by the immune system, favored an increase in specific types of white blood cell that kill tumor cells, and inhibited secretion of PGE2, an inflammatory substance in the tumor microenvironment that interferes with the anti-tumor action of certain defense cells.
“Although these studies involved animals, the results are highly promising and point to the possibility of studies in humans, as the compounds used in the nanomicelles have been approved for human use,” said Faccioli, who conducted postdoctoral research at the National Heart and Lung Institute, Imperial College London (UK).
Besides USP and Bajaj’s lab, Amity Institute of Integrative Sciences and Health (Haryana state), the Department of Surgical Oncology at All India Institute of Medical Sciences (New Delhi), the National Institute of Immunology (New Delhi) and the National Institute of Biomedical Genomics (Kalyani, West Bengal state) also took part in the research.
More information:
Poonam Yadav et al, Engineered nanomicelles inhibit the tumour progression via abrogating the prostaglandin-mediated immunosuppression, Journal of Controlled Release (2024). DOI: 10.1016/j.jconrel.2024.03.009
Citation:
Chimeric nanomicelles show promise as alternative treatment for solid tumors (2024, June 6)
retrieved 7 June 2024
from
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.
link